

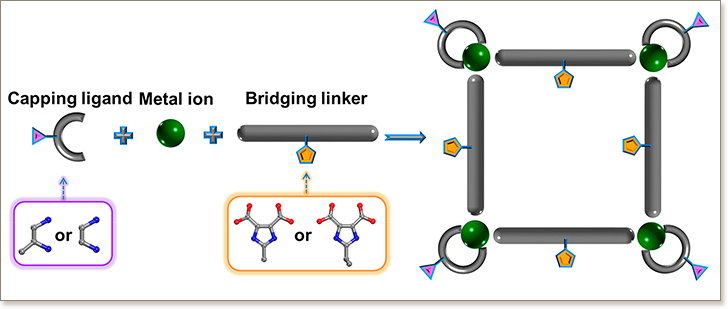
A strategy based on metal-ligand directed assembly of metal-organic squares (MOSs), built-up from four-membered ring (4MR) secondary building units (SBUs), has been employed for the design and construction of isoreticular zeolite-like supramolecular assemblies (ZSAs). Four porous Co-based ZSAs having the same underlying gis topology, but differing only with respect to the capping and bridging linkers, were successfully isolated and fully characterized. In this series, each MOS in ZSA-3-ZSA-6 possess an ideal square geometry and is connected to four neighboring MOS via a total of 16 hydrogen bonds to give a 3-periodic porous network.To systematically assess the effect of the pore system (size and functionality) on the gas adsorption properties, we evaluated the MOSs for their affinity for different probe molecules such as CO2 and light hydrocarbons. ZSA-3-ZSA-6 showed high thermal stability (up to 300 °C) and was proven highly porous as evidenced by gas adsorption studies. Notably, alkyl-functionalized MOSs were found to offer potential for selective separation of CO2, C3H6, and C3H8 from CH4 and H2 containing gas stream, such as natural gas and refinery-off gases.