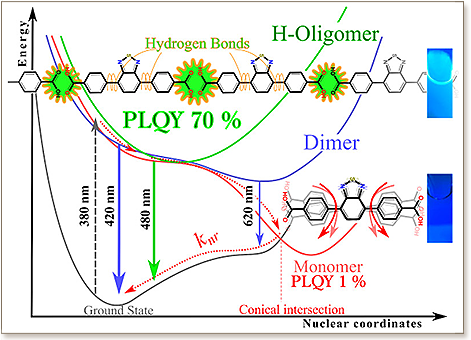
Benzothiadiazole dibenzoic acid (BTDB) derivative is a well-known organic linker in various metal–organic framework structures as well as a fluorescent probe in biological systems. Here, we demonstrate that the radiative and nonradiative decay channels of BTDB can be interplayed and precisely controlled through concentration and hydrogen-bond interactions as directly evidenced experimentally and theoretically. This leads to excited-state structural changes that significantly suppress the torsional motion around the benzothiadiazole moiety, leading to an enormous increase in the emission quantum yields from ∼1 to 70%. These changes are associated with the existence of two equilibria, where dimers and small oligomers form in dimethylformamide (DMF), with high formation constants of 18 000 M–1 and 1.2 × 1013 M–3, respectively. These evolving species, i.e., the dimers and oligomers, are formed via hydrogen bonds between carboxylic acid groups present at the far edge of the rodlike BTDB molecules. The estimated repeating number for this small-oligomer formation via bonded monomers is eight in DMF, as shown by emission spectra analysis. With deprotonation as a control experiment, these associated species can easily collapse with the initial monomer species, confirming the role of the hydrogen-bond formation in the observed phenomena. Theoretical studies and NMR experiments not only confirm the existence of the dimers, but also demonstrate the important role of the hydrogen bonds in the excited-state dynamics. These new findings provide a better understanding of the photophysical behaviors of organic linkers used in a wide range of chemical and biological applications.